Teaching my 14–16 students, I won’t go into the full details of subshells and orbitals (unless they are particularly keen). How these orbitals overlap when electrons are shared in covalent bonds dictates the strength and type of covalent bonds formed. For example, s orbitals are generally spherical and p orbitals look a bit like dumb-bells. The particular shapes of the orbitals, shown in the figure above, directly impact the shapes and properties of molecules. At scandium, the relative energy of the 3d and 4s orbitals are reversed, and one of its 21 electrons goes in a 3d orbital. However, in potassium and calcium atoms, the 3d orbitals are higher in energy than the 4s, so the latter orbitals fill with electrons first. As the electrons fill the 3s and 3p orbitals, we account for sodium through to argon. There are complex rules explaining the order of the filling of the orbitals. The third shell is formed of one 3s orbital, three 3p orbitals and five 3d orbitals. This accounts for the electronic structures of beryllium (2,1 or 1s 22s 1) through to neon (2,8 or 1s 22s 22p 6). The second shell is formed of an s-type subshell (one orbital, hence two electrons, called 2s) and a p-type subshell (three orbitals, hence six electrons, called 2p). This accounts for the electronic structure of hydrogen (1 or 1s 1) and helium (2 or 1s 2). Two electrons with opposite spin can exist in this orbital. For example, the first shell is formed of one s-type subshell, made up of one orbital that we call 1s. Within each orbital, two electrons can exist.Įach shell has one more subshell than the previous shell, and each subshell has two more orbitals then the previous subshell. These quantum numbers help to describe the electron shells, which have a complex substructure, being made of subshells, which themselves are made of orbitals. Using the rules of quantum mechanics, we can derive four numbers which identify the position and energy of electrons in atoms. Electrons have both particle-like and wave-like properties. While we usually consider electrons to be particles, they are more accurately described as quantum particles. The reason for this apparent anomaly becomes clear when we look at the more sophisticated model used for post-16 chemistry. At calcium (20 electrons, so 2,8,8,2) we hit the limit of this model, as the electronic structure of scandium (21 electrons) is 2,8,9,2, rather than the expected 2,8,8,3 from the model we use. We often say there are a maximum of two electrons in the first shell, and eight in the subsequent shells. Why do we stop at calcium when describing electronic structures at 14–16? This is a direct consequence of the simplified model we use. 
why zinc isn’t really a transition element.why transition metals are highly coloured.Ideas that can’t be explained using the 14–16 model of electronic structure include: 
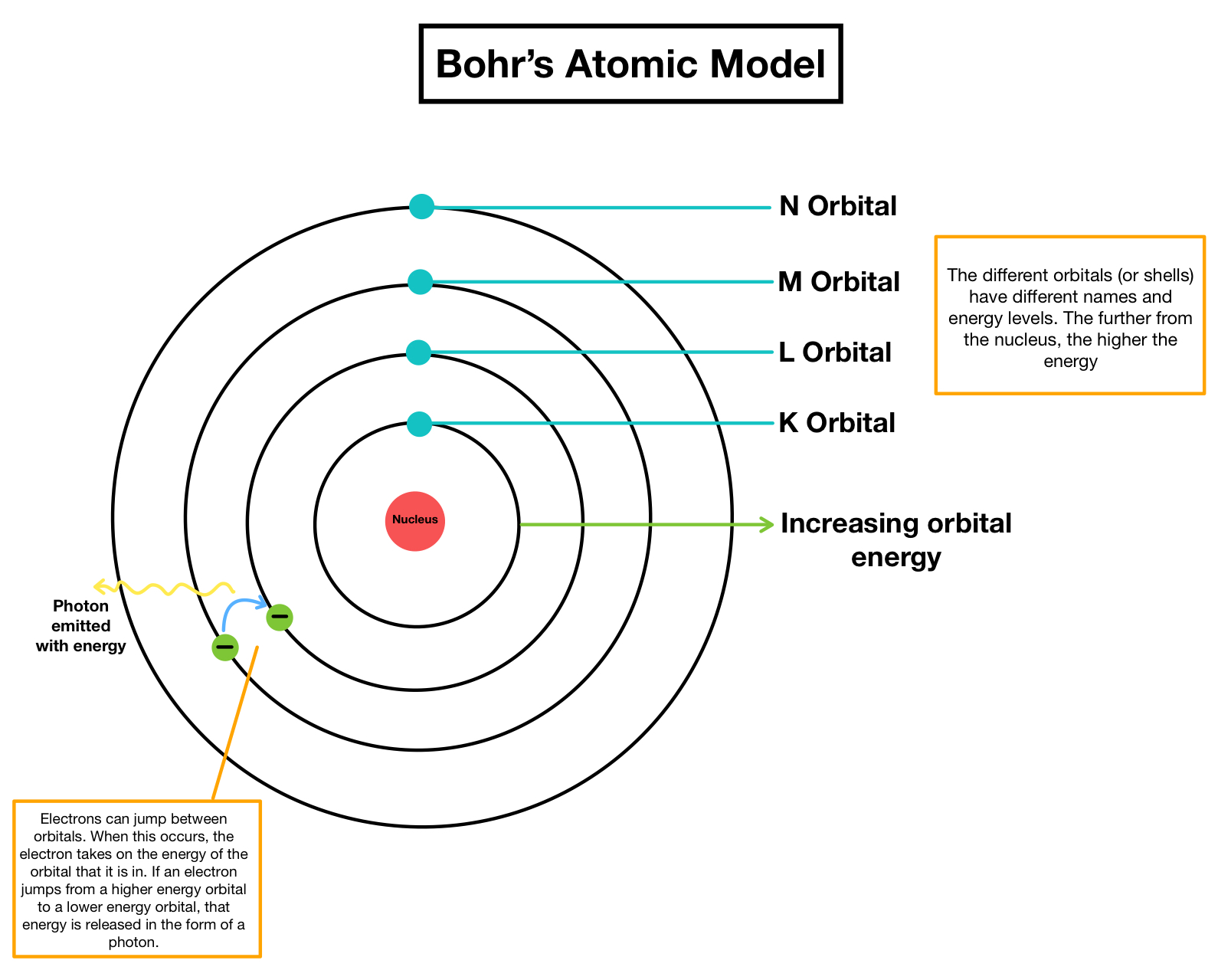
Potential issues with the model we use come from students wondering ‘what are the shells made from?’, and the inability of the model to account for the three-dimensional shape of simple molecules. Bohr, in his interpretation of atomic spectra, proposed that electrons exist in ‘orbits’ (now shells) with fixed energy levels, and that electrons can move between the orbits when energy is absorbed or emitted. 
Rutherford inferred the existence of atomic nuclei from the gold foil experiments carried out by Geiger and Marsden. This model primarily stems from the work of Hans Geiger, Ernest Marsden, Ernest Rutherford and Niels Bohr. Understanding the post-16 models is useful when teaching 14–16 students, to minimise student misconceptions.Īt 14–16, we model the atom as a tiny, massive, positively-charged nucleus, surrounded by shells of electrons. The models used at 11–14, 14–16 and post-16 become increasingly more complex. Your 14–16 learners don’t need to know that the shapes of orbitals impact the shapes and properties of molecules, but it can help them to appreciate why we only teach them certain elements’ electronic structuresĭescribing the structure of atoms relies on models. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
